Olpidium virulentus
Introduction
Olpidium virulentus is a parasitic fungus that infects plant roots, including leafy greens grown hydroponically. The pathogen can transmit two serious viruses: Lettuce big-vein associated virus (LBVaV) and Mirafiori Lettuce Big Vein Virus (MLBBV), causing a decrease in overall yield. Olpidium virulentus does not cause significant plant diseases but is a vector for viruses that can lead to infections. However, a recent report from Japan showed that Olpidium virulentus caused damage to spinach roots on a hydroponic farm. You can read more about it from the Annual Report for Plant Protection (Kansai Region): https://www.jstage.jst.go.jp/article/kapps/62/0/62_157/_pdf#:~:text=virulentus%20infects%20and%20propagates%20in,zoospores%20through%20the%20nutrient%20water.


Most of the time, the direct impact of Olpidium virulentus on plants is minimal. But, its role as a vector for plant viruses makes it a significant concern for growers. When the fungus infects plant root cells and reaches maturity, zoospores with single flagella are released from the root cell to the environment. It uses the flagella to propel themselves further, unlike spores that cannot move by themselves [3]. Therefore, they have a significant advantage in spreading water or the water phase of the soil. Olpidium virulentus is a constant threat to those growing leafy greens in hydroponic systems, as the water is the optimal place for the swimming spores of the fungus.
Other Olpidium species
It’s important to distinguish between Olpidium virulentus and Olpidium brassicae as two different species. Olpidium virulentus can infect many plants except for the mustard/cabbage family (Brassicaceae) and spread plant viruses, while Olpidium brassicae primarily infects Brassicaceae plants but can also infect other plants. Scientists have confirmed these differences through molecular analysis and host range studies [4]. When planning to test for pathogens, ensure to check which organism you are targeting.
Table of Vector, Virus, and Hosts
| Olpidium species | Virus transmitted | Acronym | Host plant |
|
O. bornovanus
|
Cucumber necrosis virus | CNV | Cucumber |
| Cucumber leaf spot virus | CLSV | Cucumber | |
| Cucumber soil-borne virus | CSBV | Cucumber | |
| Melon necrotic spot virus | MNSV | Melon | |
| Squash necrosis virus | SqNV | Squash | |
| Red clover necrotic mosaic virus | RCNMV | Red clover | |
|
O. brassicae
|
Chenopodium necrosis virus | ChNV | Chenopodium |
| Lisianthus necrosis virus | LNV | Lisianthus | |
| Tobacco necrosis virus-A | TNV-A | Tobacco | |
| Tobacco necrosis virus-D | TNV-D | Tobacco | |
| Tulip mild mottle mosaic virus | TMMMV | Tulip | |
| Freesia sneak virus | FreSV | Freesia | |
| Lettuce ring necrosis virus | LRNV | Lettuce | |
| Olive mild mosaic virus | OMMV | Olive | |
|
O. virulentus
|
Mirafiori lettuce big-vein virus | MLBVV | Lettuce, Sonchus hydrophyllus, Actites sp.* |
| Lettuce big vein associated virus | LBVaV | Lettuce, S. hydrophyllus* | |
| Pepino mosaic virus | PepMV | Tomato | |
| Tobacco stunt virus | TStV | Tobacco |
Table from Maccarone (2013), https://doi.org/10.1094/PDIS-10-12-0979-FE
We provide great overviews of many agricultural microorganisms. Subscribe to stay updated!
Symptoms
The symptoms of Olpidium virulentus with transmitted viruses in plants include chlorophyll clearing, necrosis, and reduced shoot and root growth [5]. In lettuce, the infection by LBVaV and MLBBV can lead to the development of large vein and ring necrosis diseases, which can cause significant yield losses [1]. Olpidium virulentus is reported to induce browning of the roots, growth delay, and overall yield reduction in hydroponic spinach [6].


Life Cycle and Infection Stages
The life cycle of Olpidium virulentus consists of three developmental stages: zoospores, zoosporangia, and resting spores [7]. The fungus infects the root surface (cells and root hairs) by penetrating the cell wall and developing as unicellular zoosporangia [8]. The zoosporangia then produce zoospores with single posterior flagella. These are liberated from the root cell through spore exit tubes [8]. If the fungus chooses to go dormant, it will produce resting spores and thick walls, which can survive in the environment for extended periods. The high density of zoospores in drainage water can increase the transmission of the pathogen in hydroponic systems [7].
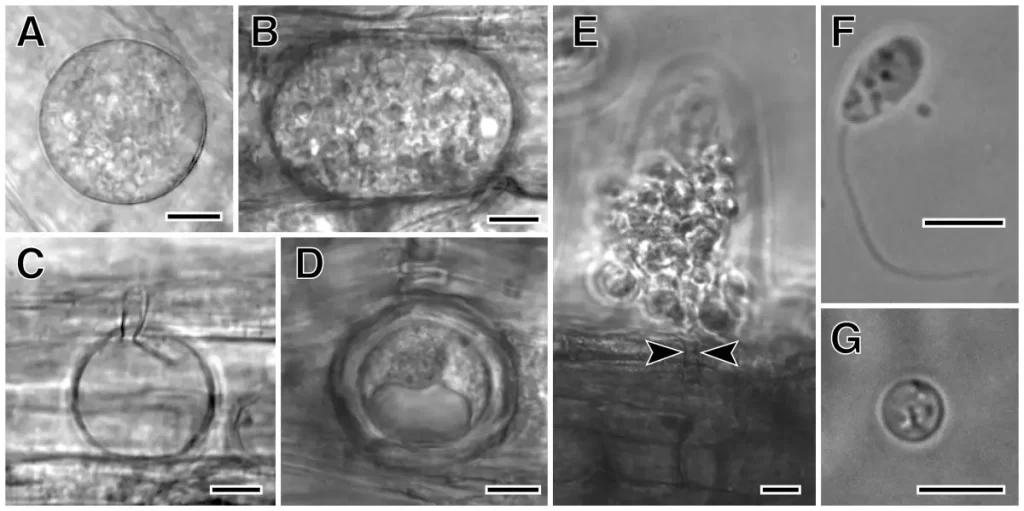
Growth Conditions for Olpidium virulentus
Olpidium virulentus is widespread worldwide and can infect plants grown in soil and hydroponic systems [1]. The fungus thrives in soilless cultures, where it finds favourable conditions for its zoospore stage to swim, spread and infect other roots [5]. In soil, the zoospores are motile when water is present, and the resting spores can survive more prolonged periods.
Methods of Prevention and Control
No curative control method can effectively control the viruses transmitted by Olpidium virulentus during cultivation [89]. However, several preventive measures can be taken to minimize the risk of infection:
- Use healthy plants and seeds to avoid introducing the pathogen into the growing system [8].
- Remove and destroy plant debris, particularly root systems, to prevent the spread of resting spores [9].
- Implement proper soil/media/substrate preparation and drainage in future growing plots [9].
- Monitor zoospore activity in water to reduce the risk of transmission in hydroponic systems [7].
Conclusion
Olpidium virulentus significantly threatens leafy greens, particularly lettuce, grown in hydroponic systems. By understanding the pathogen’s life cycle, symptoms, and growth conditions, growers can implement preventive measures to minimize the risk of infection and protect their crops. Continuous monitoring and management of zoospore activity in water are crucial to ensure the success of hydroponic cultivation.
Disclaimer
The information we present in Pathogen Profile is based on collating published peer-reviewed scientific literature, and sources we think are reliable. This is by no means an exhaustive review of pathogens. Pathogen Profile gives a small glimpse of what is known about pathogens. We encourage growers to do more research on the pathogens concerning their crops and hydroponic systems. We are not plant pathologists; thus, the information presented in the Pathogen Profile should not be used as professional advice to treat pathogens or operate your system.
References
[1] Campbell, L. G., & Madden, L. V. (2010). A comparison of Olpidium isolates from a range of host plants using internal transcribed spacer sequence analysis and host range studies. Mycological Research, 114(1), 26-35. https://www.sciencedirect.com/science/article/abs/pii/S0953756209001865
[2] Ephytia. (n.d.). Salads – Olpidium virulentus (Olpidium brassicae, Fungal vector virus). http://ephytia.inra.fr/en/C/5421/Salads-Olpidium-virulentus-Olpidium-brassicae-Fungal-vector-virus
[3] Maccarone, L. D. (2013). Relationships between the pathogen Olpidium virulentus and viruses associated with lettuce big-vein disease. Plant Disease, 97(5), 700-707. https://doi.org/10.1094/PDIS-10-12-0979-FE
[4] Lay, C.-Y., Hamel, C., & St-Arnaud, M. (2018). Taxonomy and pathogenicity of Olpidium brassicae and its allied species. Fungal Biology, 122(9), 837-846. https://doi.org/10.1016/j.funbio.2018.04.012
[5] Ephytia. (n.d.). Tomato – Olpidium brassicae (Root fungus). http://ephytia.inra.fr/en/C/5011/Tomato-Olpidium-brassicae-Root-fungus
[6] Nishimura, S., Kusakari, S., & Tojo, M. (2020). Effects of fungicides and antifungal agents on Olpidium root rot of spinach in a hydroponic culture. Ann. Rept. Kansai Pl. Prot., 62, 157-159. https://www.jstage.jst.go.jp/article/kapps/62/0/62_157/_pdf#:~:text=virulentus%20infects%20and%20propagates%20in,zoospores%20through%20the%20nutrient%20water
[7] Alfaro-Fernández, A., Córdoba-Sellés, M. D. C., Herrera-Vásquez, J. Á., Cebrián, M. D. C., & Jordá, C. (2010). Transmission of Pepino mosaic virus by the fungal vector Olpidium virulentus. Journal of Phytopathology, 158(4), 217-226. https://doi.org/10.1111/j.1439-0434.2009.01605.x
[8] Sekimoto, S., Rochon, D., Long, J. E., Dee, J. M., & Berbee, M. L. (2011). A multigene phylogeny of Olpidium and its implications for early fungal evolution. BMC Evolutionary Biology, 11, 331. https://doi.org/10.1186/1471-2148-11-331
[9] Ephytia. (n.d.). Salads – Protection methods. http://ephytia.inra.fr/en/C/6022/Salads-Protection-methods
![]()
David Santos is the COO/CMO of Healthy Hydroponics InnoTech





